For example, frozen nitrogen will form both the liquid phase and the vapor phase when exposed to normal temperature and pressure. In case of liquid to gas phase change, this amount of energy is known as the enthalpy of vaporization, (symbol H vap unit: J) also known as the (latent) heat of vaporization or heat of. 
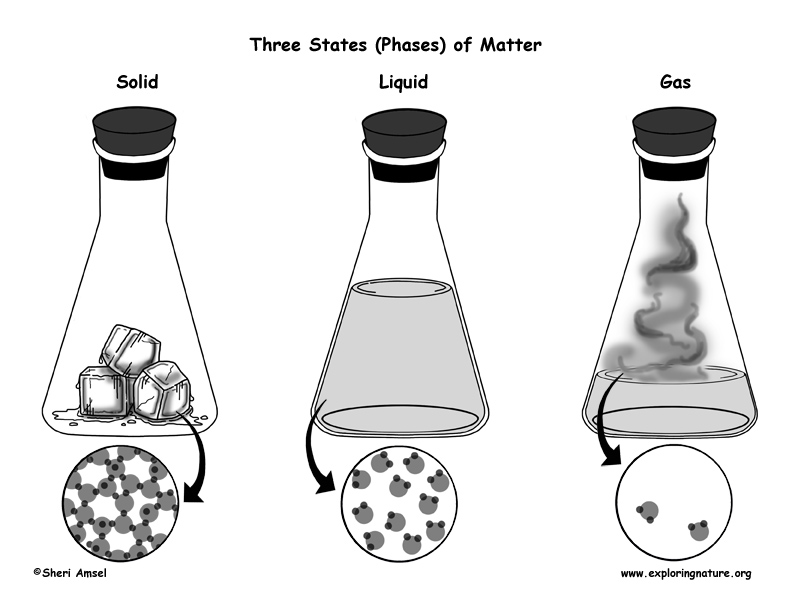
Multiple phase changes can occur at once. In general, when a material changes phase from solid to liquid, or from liquid to gas a certain amount of energy is involved in this change of phase. For example, if you view the sublimation of dry ice into carbon dioxide gas, the white vapor that is observed is mostly water that is condensing from water vapor in the air into fog droplets. Phase changes aren't always clear when observing a situation. Plasma most often forms from ionization of a gas, although if sufficient energy and enough space are available, it's presumably possible for a liquid or solid to ionize directly into a gas. Plasma: Plasma can recombine to form a gas. Video Transcript Solids, liquids and gases. Gases form from the sublimation of solids, vaporization of liquids, and recombination of plasma. particles are arranged in solids, liquids and gases. Gases: Gases can ionize into plasma, condense into liquids, or undergo deposition into solids. Liquids form by condensation of gases and melting of solids. Liquids: Liquids can vaporize into gases or freeze into solids. The enthalpy change during melting or fusion is the latent heat. 
The latent heat of fusion of ice is 3.34 x 10 joules per kilograms (or, 3.34 x 10J/Kg). A phase of a thermodynamic system and the. Commonly the term is used to refer to changes among the basic states of matter: solid, liquid, and gas, and in rare cases, plasma. Solids form by deposition from gases or freezing of liquids. The latent heat of fusion or melting, of a solid, is the quantity of heat in joules required to transform a solid, at its melting point, to a liquid without any variation in temperature. In chemistry, thermodynamics, and other related fields, a phase transition (or phase change) is the physical process of transition between one state of a medium and another. Solids: Solids can melt into liquids or sublime into gases. Another way to list phase changes is by states of matter: 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
